When does entropy increase8/31/2023  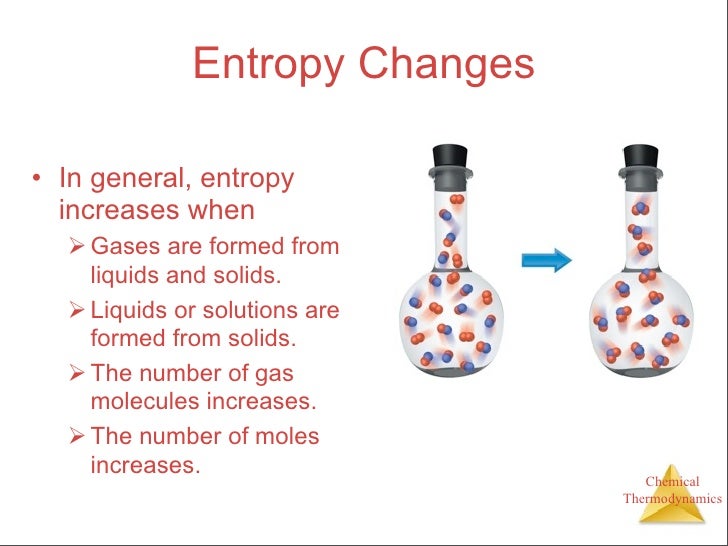 
rather, it's more of a statistical observation. A paper that my professor once showed me makes it clear that entropy isn't a measure of energy density/distribution. I'm having a difficult time understanding how we can relate the idea of entropy to energy. But this entropy cost is offset by a greater decrease in enthalpy, bringing the overall $ΔG$ down (so it's negative), and therefore making the bond "stabilizing". However, when hydrogen bonds form, the $ΔS$ actually decreases there is more order when molecules align to form H-bonds. 
We call the formation of hydrogen bonds as being "stabilizing" - that is, they lower the overall energy $ΔG$. Now, before I explain my confusion, I'd like to share an example from my course: hydrogen bonding (non-covalent interactions. There are many formulas that relate entropy to energy. However, in my understanding, the idea is still the same: increasing entropy is really just a statistical likely-hood that will hold true on average. Of course there's a more elaborate definition involving macrostates and microstates, where entropy is greater when a macrostate has more microstates. Therefore, on average, they will spread around and entropy is increased. For example, if you have a vacuum inside of a box and you place a handful of gas atoms on one side, the molecules have a higher statistical probability to spread out, versus remain in one concentrated spot. Conceptually, I've always understood entropy to be a statistical idea. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
